 Invest in BioXgen
Invest in BioXgen Invest in BioXgen
Invest in BioXgen
 Invest in BioXgen
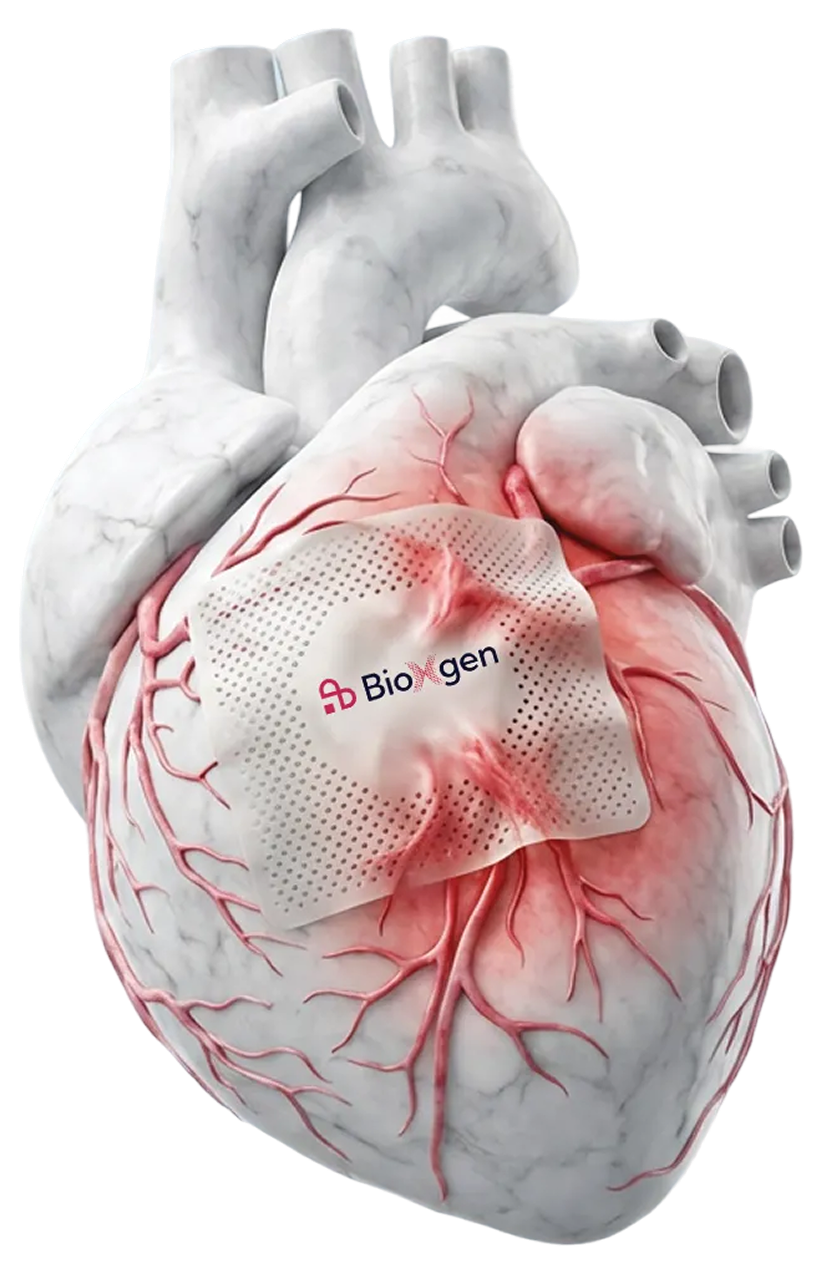
Invest in BioXgenBioXgen’s patent-pending ExtraCellular Matrix (ECM)-based cardiac patch supports patients’ recovery after open-heart surgery. Post-surgery complications such as post-operative atrial fibrillation (POAF) can affect over 50% of cardiac surgery patients within 3-4 days after surgery. We are on an accelerated track for FDA use clearance in 2026. This raise expedites the path from clinical trials to market entry, positioning us to address post-operative surgery complications, improve patient outcomes while saving hospitals and healthcare payers billions each year.

The founders have an established history of success from start-ups to acquisition valued in total at ~$65M.
.svg)
Up to 50% of cardiac surgery patients develop POAF, the #1 postoperative complication in this patient population.
.svg)
500,000 open-heart surgeries performed annually in the U.S., representing approximately $9 billion in POAF-related costs.
.svg)
510(k) predicate device confirmed, providing a clear regulatory route to market that typically takes less time than de novo approval.
.svg)
POAF can cost hospitals $10,000 to $20,000 per patient through extended ICU stays and readmissions.
.svg)
ECM-based cardiac patch with intellectual property protection covering material composition and application method.
.svg)
Preclinical results completed, with partnerships in place at leading U.S. cardiac surgery centers for first-in-human trials.
.svg)
Founded by an MD/PhD CEO with a background in medical pharmacology / new drug discovery and prior company-building experience, alongside a PhD Chief Science Officer- a Professor of Physiology with 30+ years in cardiac physiology, biomedical engineering, and translational research.
.svg)
Post-operative complication management has become prioritized. Modern Healthcare systems use value-based medical models that incentivize risk mitigation and reward improved patient outcomes.
.svg)
BioXgen operates in cardiovascular medtech, a sector where successful products have historically led to major strategic acquisitions following regulatory clearance.
.svg)
Founded by an MD/PhD CEO with a background in medical pharmacology / new drug discovery and prior company-building experience, alongside a PhD Chief Science Officer- a Professor of Physiology with 30+ years in cardiac physiology, biomedical engineering, and translational research.

Every day, cardiac surgery teams face a predictable problem. Despite advances in surgical technique, up to half of their open-heart patients develop POAF. For hospitals operating under fixed reimbursement models, each case represents thousands in uncompensated costs. This cost extends to healthcare payers after hospitalization.
BioXgen developed an ECM-based cardiac patch to address this.

Derived from porcine tissue applied directly to the heart’s surface during surgery

Uses porcine ECM material with 100+ years of proven safety in human use

Designed specifically for cardiac applications

Seamlessly integrates into existing surgical workflows, requiring no new equipment or additional training
Our ECM-based device is designed to provide structural support for the heart during the critical postoperative recovery period.
Our product sits at the intersection of cardiovascular devices and regenerative biomaterials. We designed it for everyday cardiac teams, not research labs. With our regulatory pathway confirmed and clinical partnerships established, we’re positioned to bring this technology through validation and into operating rooms where it can make a difference.
Postoperative atrial fibrillation remains the most common complication following cardiac surgery.
The clinical consequences are severe:
2x higher death rate compared to patients without POAF

4x increase in stroke incidence

3 to 7 additional days in the ICU

2x higher death rate compared to patients without POAF

~45% develop recurrent Afib

Current interventions remain limited and inconsistent, leaving surgical teams without reliable tools to support their patients during recovery.
BioXgen’s patent-pending ECM-based patch is designed to support cardiac surgery recovery. The device features:
.svg)
Porcine tissue-derived ECM
.svg)
Epicardial application
.svg)
Surgical workflow integration
.svg)
Natural material
.svg)
Sustainability
.svg)
Scalability
The material is designed to be absorbed as the tissue heals, leaving no permanent foreign material.
The market for POAF management aligns with fundamental healthcare economics. Cardiac surgery represents one of the most common and expensive procedures in modern medicine.

BioXgen has laid the foundation for advancing from preclinical development to clinical trials.

.svg)
We've confirmed the regulatory route for clearance, providing clarity on requirements and timeline
.svg)
Over 200 patients undergoing cardiac surgery received a similar treatment with a human-derived placental (amnion) ECM patch

.svg)
We're preparing for first-in-human studies, the critical step that will generate the clinical data needed for FDA submission. Our partnerships with cardiac surgery centers provide access to patient populations and the surgical expertise required for successful trials.

.svg)
Agreements in place with leading cardiac surgery centers for first-in-human trials
.svg)
Early discussions with cardiac surgeons confirm adoption readiness and workflow compatibility

.svg)
Founded by an MD/PhD CEO with a background in medical pharmacology / new drug discovery and prior company-building experience, alongside a PhD Chief Science Officer who is a Professor of Physiology with 30+ years in cardiac physiology, biomedical engineering, and translational research.
.svg)
Patent-pending intellectual property covering both the patch material composition and application methodology
The material is designed to be absorbed as the tissue heals, leaving no permanent foreign material.
.svg)
Derived from porcine tissue applied directly to the heart’s surface during surgery
.svg)
Pricing designed to deliver clear ROI through complication management and associated cost savings
.svg)
Per-procedure pricing model aligns with existing hospital purchasing and inventory systems
Once we receive FDA clearance, we’ll target cardiac surgery centers through a combination of direct sales and established medical device distribution channels. The value proposition centers on demonstrated cost savings, making the economic case straightforward for hospital decision-makers.
Hospital purchasing committees evaluate devices based on clinical evidence, cost-effectiveness, and ease of implementation. Our design specifically addresses these criteria by integrating into existing workflows, requiring minimal training, and targeting a complication with clear cost implications.
This raise supports the critical path to FDA clearance and market entry.

.svg)
First-in-human studies, data collection, FDA submission preparation, and regulatory consulting
.svg)
Refining manufacturing processes, scaling production capabilities, and quality systems implementation
Clinical trials are our primary capital deployment. The first-in-human studies will generate the safety and efficacy data required for an FDA 510(k) submission. This includes patient enrollment, surgical procedure costs, follow-up monitoring, and data analysis.
Manufacturing scale-up ensures we can produce the cardiac patch at volumes needed for clinical trials while establishing the quality systems and documentation the FDA requires for commercial production.
Postoperative atrial fibrillation has been accepted as an inevitable complication of cardiac surgery for too long. Thousands of patients each year face doubled mortality risk, extended ICU stays, and increased stroke danger because of POAF.
BioXgen’s ECM-based cardiac patch is designed to support the recovery process. By providing structural support during the critical postoperative period, we aim to help cardiac surgery teams support better patient outcomes.
The next phase requires clinical trials to evaluate this approach in real cardiac surgery patients.
.svg)
Bonus Units: 1%
.svg)
Bonus Units: 3%
.svg)
Bonus Units: 5%
.svg)
Bonus Units: 7%
.svg)
Bonus Units: 10%
.svg)
Bonus Units: 15%
.svg)
Bonus Units: 20%
.svg)
Bonus Units: 3%
.svg)
Bonus Units: 5%
.svg)
Bonus Units: 7%
.svg)
Bonus Units: 10%
.svg)
Bonus Units: 15%
.svg)
Bonus Units: 20%
.svg)
Bonus Units: 25%
.svg)
Regulation CF allows investors to invest in startups and early-growth companies. This is different from helping a company raise money on Kickstarter; with Regulation CF Offerings, you arenʼt buying products or merchandise – you are buying a piece of a company and helping it grow.
.svg)
Accredited investors can invest as much as they want. But if you are NOT an accredited investor, your investment limit depends on either your annual income or net worth, whichever is greater. If the number is less than $124,000, you can only invest 5% of it. If both are greater than $124,000 then your investment limit is 10%.
.svg)
To calculate your net worth, just add up all of your assets and subtract all of your liabilities (excluding the value of the personʼs primary residence). The resulting sum is your net worth.
.svg)
We cannot give tax advice, and we encourage you to talk with your accountant or tax advisor before making an investment.
.svg)
Individuals over 18 years of age can invest.
.svg)
There will always be some risk involved when investing in a startup or small business. And the earlier you get in the more risk that is usually present. If a young company goes out of business, your ownership interest could lose all value. You may have limited voting power to direct the company due to dilution over time. You may also have to wait about five to seven years (if ever) for an exit via acquisition, IPO, etc. Because early-stage companies are still in the process of perfecting their products, services, and business model, nothing is guaranteed. Thatʼs why startups should only be part of a more balanced, overall investment portfolio.
.svg)
The Common Stock (the “Shares”) of BioXgen (the “Company”) are not publicly-traded. As a result, the shares cannot be easily traded or sold. As an investor in a private company, you typically look to receive a return on your investment under the following scenarios: The Company gets acquired by another company. The Company goes public (makes an initial public offering). In those instances, you receive your pro-rata share of the distributions that occur, in the case of acquisition, or you can sell your shares on an exchange. These are both considered long-term exits, taking approximately 5-10 years (and often longer) to see the possibility for an exit. It can sometimes take years to build companies. Sometimes there will not be any return, as a result of business failure.
.svg)
Shares sold via Regulation Crowdfunding offerings have a one-year lockup period before those shares can be sold under certain conditions. The exceptions are sales to:
(i) to the Company;
(ii) to an “accredited investorˮ within the meaning of Rule 501 of Regulation D under the Securities Act;
(iii) as part of an offering registered under the Securities Act with the SEC; or
(iv) to a member of the Investorʼs family or the equivalent, to a trust controlled by the Investor, to a trust created for the benefit of a member of the family of the Investor or equivalent, or in connection with the death or divorce of the Investor or other similar circumstance.
.svg)
In the event of death, divorce, or similar circumstance, shares can be transferred to:
The company that issued the securities
An accredited investor
A family member (child, stepchild, grandchild, parent, stepparent, grandparent, spouse or equivalent, sibling, mother-in-law, father-in-law, son-in-law, daughter-in-law, brother-in-law, or sister-in-law, including adoptive relationships)
.svg)
If a company does not reach their minimum funding target, all funds will be returned to the investors after the close of the offering.
.svg)
All available disclosure information can be found on the landing pages for our Regulation Crowdfunding offering.
.svg)
You can cancel your investment at any time, for any reason, until 48 hours prior to a closing occurring. If youʼve already funded your investment and your funds are in escrow, your funds will be promptly refunded to you upon cancellation. To submit a request to cancel your investment please email: info@dealmakersecurities.com
.svg)
At a minimum, the company will be filing with the SEC and posting on itʼs website an annual report, along with certified financial statements. Those should be available 120 days after the fiscal year end. If the company meets a reporting exception, or eventually has to file more reported information to the SEC, the reporting described above may end. If these reports end, you may not continually have current financial information about the company.
.svg)
DealMaker Securities is serving as the intermediary for this offering. Once an offering ends, there is no guarantee that DealMaker Securities will have a relationship with the company. The company may continue its relationship with DealMaker Securities for additional offerings in the future. DealMaker Securitiesʼ affiliates may also provide ongoing services to the company. There is no guarantee any services will continue after the offering ends.
Equity crowdfunding investments in private placements, and start-up investments in particular, are speculative and involve a high degree of risk and those investors who cannot afford to lose their entire investment should not invest in start-ups. Companies seeking startup investment through equity crowdfunding tend to be in earlier stages of development and their business model, products and services may not yet be fully developed, operational or tested in the public marketplace. There is no guarantee that the stated valuation and other terms are accurate or in agreement with the market or industry valuations.
Further, investors may receive illiquid and/or restricted stock that may be subject to holding period requirements and/or liquidity concerns.
Forward looking statements were included here that the Company believes to be accurate given the current information. They involve known and unknown risks, uncertainties and other important factors which if changed may affect the outcome(s).
DealMaker Securities LLC, a registered broker-dealer, and member of FINRA | SIPC, located at 30 East 23rd Street, 2nd Floor, NY, NY 10010, is the Intermediary for this offering and is not an affiliate of or connected with the Issuer. Please check our background on FINRA’s BrokerCheck.
DealMaker Securities LLC does not make investment recommendations.
DealMaker Securities LLC is NOT placing or selling these securities on behalf of the Issuer.
DealMaker Securities LLC is NOT soliciting this investment or making any recommendations by collecting, reviewing, and processing an Investor’s documentation for this investment.
DealMaker Securities LLC conducts Anti-Money Laundering, Identity and Bad Actor Disqualification reviews of the Issuer, and confirms they are a registered business in good standing.
DealMaker Securities LLC is NOT vetting or approving the information provided by the Issuer or the Issuer itself.
Contact information is provided for Investors to make inquiries and requests to DealMaker Securities LLC regarding Regulation CF in general, or the status of such investorʼs submitted documentation, specifically. DealMaker Securities LLC may direct Investors to specific sections of the Offering Circular to locate information or answers to their inquiry but does not opine or provide guidance on issuer-related matters.
Forward looking statements were included here that the Company believes to be accurate given the current information. They involve known and unknown risks, uncertainties and other important factors which if changed may affect the outcome(s).
Past performance is not indicative of future results.
©2026 All Rights Reserved